3.2.3 Left ventricular function
3.2.3.1 Pathophysiology of left ventricular dysfunction
The most common reason for referral to echocardiography is left ventricular function. Assessment of left ventricular function is extremely important. It correlates with symptoms, prognosis, events, and complications in a large number of conditions. We base many decisions in cardiology on left ventricular function. However, proper comprehension of left ventricular function requires that we understand a few fundamental principles. Left ventricular function is usually established by computing or estimating ejection fraction. Ejection fraction is the percentage of blood ejected from the ventricle during systole in relation to the total end-diastolic volume. Visually we judge left ventricular function on the basis of how much smaller the ventricle becomes during systole. When the function of the heart is impaired, less blood will be ejected and the ejection fraction will fall. Ejection fraction is also a function of ventricular size. When the ventricle is large (in athletes, for instance), ejection fraction will drop. This is pure mathematics. When the ventricle is large, even a small systolic reduction in ventricular size (and a relatively low ejection fraction) will yield a sufficient stroke volume to perfuse the body. Conversely, when the ventricle is small (also known as hypovolemia), it will compensate by increasing its contractility. In this condition the ejection fraction will be higher than normal. During conventional echocardiography, left ventricular function is assessed at rest. Therefore we have no clue as to the "functional capacity" and contractile reserve of the ventricle. Obviously it would be hazardous to rely on ejection fraction alone. We will see later how this can affect our interpretation under various conditions.
In the range of normal values, the ejection fraction does not correlate with cardiovascular exercise capacity.
A reduction in ejection fraction is simply a consequence of ventricular dysfunction. When contractility drops, the heart has to compensate. This is achieved by several means:
- Elevation in sympathetic activity (which increases heart rate and contractility);
- Increase in salt and water retention (which increases blood volume and elevates preload);
- Dilatation of the ventricle - which, by the Frank starling mechanism, increases contractility. More volume is ejected relative to the degree of inward motion of the walls.
- When certain segments reveal poorer contractility, other areas will compensate by contracting "more than normal" (hypercontractility).
3.2.3.2.2 Myocardial mechanics
The left ventricle has a sophisticated orientation of fibers, which permits the heart not only to contract in radial direction, but also shorten its length. The heart contracts in its circumferential aspect as well, which results in a twisting motion.
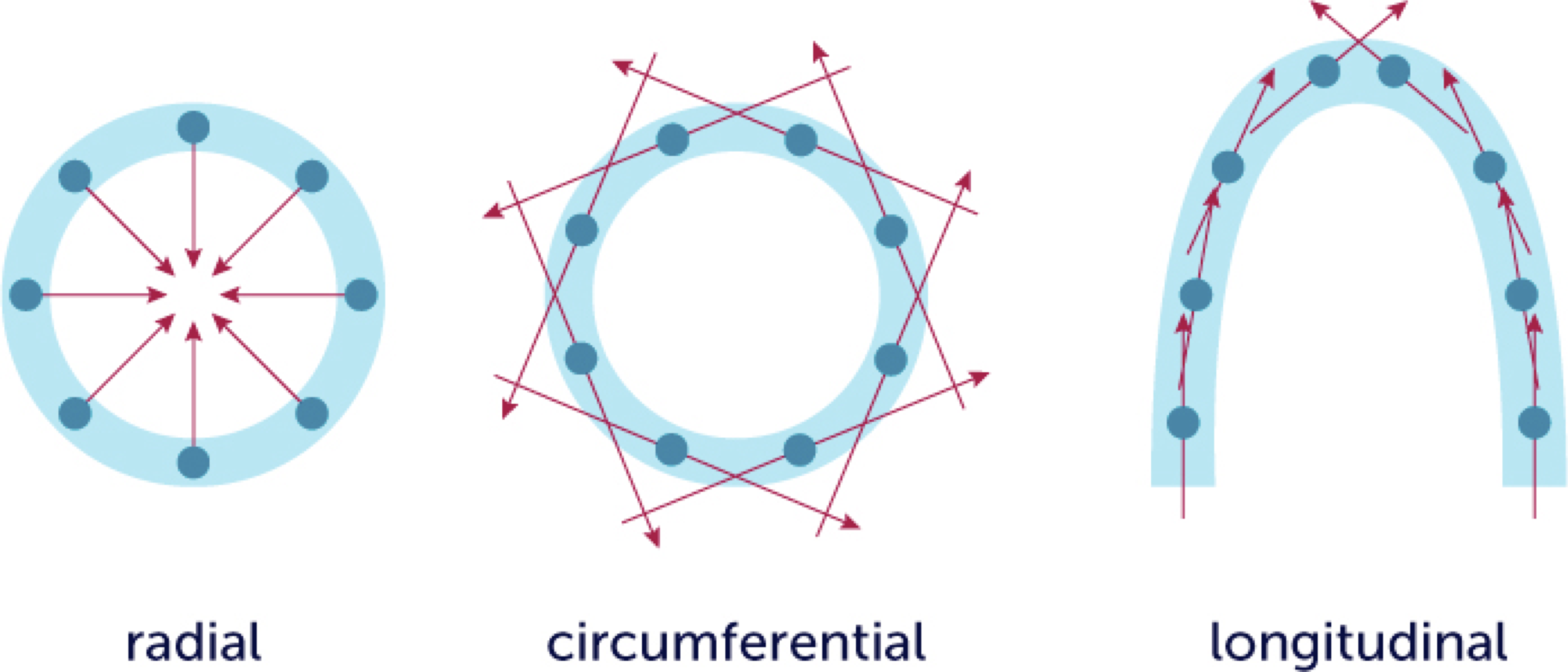
It is now a well established fact that normal functioning of these mechanics is essential for systolic as well as diastolic function, and that the mechanics of the myocardium may be disturbed in many pathologies that affect the left ventricle. Specifically, we now know that impairment of longitudinal function is a so-called early marker of left ventricular dysfunction, and that it precedes a drop in ejection fraction.
Several variables are used to describe myocardial mechanics. We can measure the velocity of the myocardium and calculate its deformation. In addition, it is possible to study the various components of contraction (radial, circumferential and longitudinal). These measurements are performed either with tissue Doppler or speckle tracking echocardiography. These techniques are described in Chapter 21, Other Techniques.
3.2.3.2 Parameters of left ventricular function
Left ventricular function is very important, but may be difficult to assess. Several methods are available, yet none is perfect. I would recommend an integrative approach using several methods. Echocardiography offers the following options:
Parameters of LV Function
- Fractional shortening
- “Eyeballing“ of LVF
- Ejection fraction (EF) - Simpson method
- Stroke volume
- Cardiac output
- Cardiac Index
- Contractility (dp/dt)
- Tei index
3.2.3.2.1 Fractional Shortening
Fractional shortening (FS) is an MMode method. You have already learned how to perform an MMode measurement through the left ventricle (see Chapter 1, How to Image). You learned that MMode provides measurements of the ventricle at end systole (LVESD) as well as end diastole (LVEDD). By entering your measurements in the following formula:
(LVEDD - LVESD / LVEDD) x 100
You will get the percentage of "size reduction" of the left ventricle. This is fractional shortening. It is simple, but remember that the value does not express ejection fraction because we are not computing volumes but distances (diameters). Fractional shortening is lower: the normal value is higher than 28% (see table below).
| male | female | |
|---|---|---|
| Normal | 25- 43% | 27- 45% |
| Mild | 20- 24% | 22- 26% |
| Moderate | 15- 19% | 17- 21% |
| Severe | ≤ 14% | ≤ 16% |
Theoretically you can also use fractional shortening to derive ejection fraction either with the cube method (LVEDV = LVEDD3, LVESV = LVESSD3) or the Teichholz formula:
End-diastolic volume=7/(2·4+EDD) x EDD3
End-systolic volume=7/(2·4+ESD) x ESD3
Compared to the cube method the Teichholz formula gives you a slightly better approximation of volumes.
Many scanners perform these calculations automatically for you. However, they are not very exact because they are based on geometric assumptions. To some extent these calculations only work for normal-sized ventricles. The error of measurement is substantial. If you wish to obtain a rough estimate of ejection fraction from fractional shortening, simply multiply the value by 2.
Do not use the Teichholz or the "cube" formula.
The fractional shortening equation is a very simple method, but has numerous limitations. One limitation has been mentioned: the diameter measurements must be correct and this is not possible when the ventricle is not cut perpendicularly by the MMode line, when the margins cannot clearly be delineated, or when you have pronounced inward motion from the lateral and septal walls in the presence of small ventricles.
In some situations it may be impossible to clearly define end diastole and end systole. This occurs in the presence of abnormal septal motion (such as dyssynchrony or pressure or volume overload of the right ventricle).
Do not use the fractional shortening method when the diameters are not representative of ventricular size. One example of this would be regional wall motion abnormalities. When a wall motion abnormality is present in the septum or posterolateral wall (the segments through which the MMode line passes), you will underestimate global left ventricular function. When there is abnormal wall motion outside the segments that can be displayed with the MMode, you will overestimate true ventricular function. To summarize, fractional shortening has limited application, cannot be used in all patients, and only provides a rough approximation of left ventricular function.
- LBBB / Dyssynchrony
- Regional wall motion abnormalities
- Poor image quality
- Abnormal septal motion
- Inadequate MMode orientation
3.2.3.2.2 The Simpson method to determine ejection fraction
The Simpson method enables us to determine the volume of the left ventricle (see section: 3.2.2.2.2). When you perform the calculation for end-diastolic as well as end-systolic volumes, you can calculate ejection fraction by using the following formula:
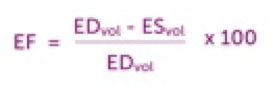
In the "monoplane Simpson method", only the four-chamber view is employed to calculate volume. In the biplane Simpson method, volume is based on the two-chamber view as well. In general the biplane Simpson method is more exact and should be given preference over the monoplane approach. Normal values of ejection fraction are shown in the following table.
| Normal | > 55 % |
| Mild | 45— 54 % |
| Moderate | 30— 44 % |
| Severe | < 30% |
Note that ejection fraction determined with echocardiography differs from that obtained with other imaging modalities. Echocardiography provides higher values than radionuclide ventriculography (RNV) or MRI. Therefore, the lower limit of normal is lower with echo.
The Simpson method is by far the most commonly used approach to quantify left ventricular function. Yet, in clinical practice you will encounter situations in which the Simpson method cannot be relied upon. Its main limitation is poor image quality, which does not permit reliable tracing of the endocardial contour (the problem of endocardial tracing has been discussed in section 3.2.2.2.2 - The Simpson method). Other limitations include dyssynchrony when it is difficult to define end-systolic and end-diastolic frames, and regional wall motion abnormalities. Remember that even when you use the biplane method you will be unable to account for wall motion abnormalities which are present in views other than the four- or two-chamber view (i.e. the posterolateral segment). The ejection fraction may also vary from beat to beat, especially in the presence of arrhythmia (i.e. atrial fibrillation). In this situation it is best to perform several measurements (something almost no investigator does!). In any case, be critical towards the values you obtain. They should always match your visual perception of left ventricular function. When the values are unreliable, simply do not use them.
Other methods have been proposed. For example, it is possible to use volume calculations based on the area length method (see section derived 3.2.2.2.1 Area Length Method). As discussed above, the method has several limitations and is therefore rarely used.
3.2.3.2.3. Three-dimensional echocardiography
Techniques that yield three-dimensional (full volume) datasets of the heart were developed to circumvent the limitations of 2D echocardiography. Three-dimensional echocardiography is performed using special transducers which are able to scan a "pyramidal" volume, often within a single beat. As the images display three dimensions of the heart as well as motion, the technology is also known as four-dimensional echocardiography. With the help of special software it is possible to reconstruct three-dimensional images of the heart and create wire frame reconstructions of the beating left ventricle. Thus, it is possible to derive global and regional volumes, volume curves, and ejection fractions.
Three-dimensional reconstruction of the left ventricle to compute global and regional ventricular function. The reconstruction displays a so-called pumping bag. The corresponding volume curves of each individual segment are shown below right:
Video Platform Video Management Video Solutions Video Player
A great deal of effort has been expended in rendering the applications as user friendly as possible. Most systems require minimal user interaction. There is convincing evidence to show that ejection fraction derived with 3D is superior to all other echocardiographic methods. The accuracy is even greater when the procedure is combined with contrast echocardiography. The technology can also be used to inspect dyssynchrony of the heart. This topic will be discussed in greater detail in the chapter on dilated cardiomyopathy. In contrast with two-dimensional echocardiography, no geometric assumptions are made. However, the problem of foreshortening persists. Besides, the frame rates (or rather volume rates) and image resolution of four-dimensional echocardiography are less than those of 2D echo. Yet, this technology is improving with time and will be integrated into more and more scanners. One may well anticipate 3D volume computations based on speckle tracking data. In any case, three-dimensional echocardiography has the potential to replace the Simpson method.
Ejection fraction derived from 3D echo is lower than that obtained with 2D echo.
get free echo lectures3.2.3.2.5 "Eyeballing" of left ventricular function
Whether visual estimation of left ventricular function is better than measuring ejection fraction is still controversially discussed. The truth is yes and no. When you are experienced, eyeballing may be better. This is especially true of poor image quality because the Simpson method has its limitations here.
How do you "eyeball" left ventricular function? Basically you should look at the following features:
- Inward motion of the endocardium
- Thickening of the myocardium
- Longitudinal motion of the mitral annulus
- Geometry of the ventricle
The following scheme may be used to grade left ventricular function:
| Hyperdynamic | > 70% |
| Normal | 69- 55% |
| Borderline | 54- 50% |
| Mildly reduced | 50- 45% |
| Mild to moderately reduced | 44- 40% |
| Moderately reduced | 39- 35% |
| Moderate to severely reduced | 34- 30% |
| Moderate to severely reduced | < 30% |
hyperdynamic:
normal:
moderately reduced:
severly reduced:
According to my experience it may be better to incorporate intermediate stages as well, such as borderline (between normal and mild), mild to moderate, and moderate to severe. In clinical practice you will be glad to have a more refined scale. Some centers directly "convert" visual estimation of left ventricular function into a value (range) for ejection fraction. (see example in the table above)
On a personal note, I have always regretted that the fact that the degree of left ventricular function is not further subdivided below 30%. After all, some patients have really poor left ventricular function of 10% or less. These (I refer to them as "disaster") ventricles probably comprise a subgroup with a very poor prognosis. It would be useful to be able to emphasize this aspect.
What are the specific issues you should consider when you "eyeball" the ventricle?
First, if you want to estimate global function you have to look at all segments. Your estimate should be based on as many segments as possible. Especially patients with regional wall motion abnormalities are difficult to assess. Some segments might be hypocontractile while others may be hyperdynamic (to compensate for the segments that cannot contribute to ejection). Thus, to derive a global estimate you should perform "mental arithmetic". You "add and subtract the function of the individual segments" (more on this issue in Chapter 8 when we deal with coronary artery disease).
Patient with a anteroseptal wall motion abnormality. The remaining segmets (Basal septum and lateral wall) is normal / hyperdynamic. Eyballing must consider all these factors.
Second, there will be situations in which certain segments will not be visible or can only be seen in individual frames. Fortunately, the eye is able to interpolate between missing data. This is one of the advantages of human evaluation as opposed to automated contour detection algorithms. Nevertheless, you need intuition, imagination and experience.
Third, the heart not only contracts, but also moves "in toto" within the chest. This is also known as total heart motion. This motion of the heart alters the cut plane and causes fluctuations in the measured volume of the ventricle during the cardiac cycle. Especially the two-chamber view is susceptible to this error. To prevent this source of error, view and record image loops of the ventricle during breathhold, and always concentrate on the apical segments. This is where this phenomenon is most prominent.
Fourth, when we assess left ventricular function on the short-axis views we are interpreting "radial" function. However, radial function is just one aspect of contraction. Therefore, also use apical views to look at "longitudinal function": observe how much the ventricle shortens in length. This is best appreciated by observing the motion of the mitral annulus. The importance of longitudinal function will be discussed in the section on deformation imaging.
In summary, "eyeballing" is not as easy as it may seem. Several factors have to be considered. Yet, generally there is fairly good agreement between observers. It is never a problem to distinguish between normal and severely reduced function
The Simpson method requires a lot of experience. Therefore, practice using the method.
Use contrast to enhance boarder delineation
3.2.3.2.6 Hemodynamic variables
Doppler echocardiography combined with 2D imaging provides numerous opportunities to derive hemodynamic information. These include stroke volume, cardiac output, and the cardiac index. All of these parameters are relevant for systolic left ventricular function and can be calculated from two simple measurements: the velocity time integral (VTI) and the cross-section of the left ventricular outflow tract. For the velocity time integral you record a PW Doppler signal from the left ventricular outflow tract (see 2.5.3 PW Doppler of left ventricular outflow tract velocity). By tracing the curve you can derive the velocity time interval (VTI), which represents the total flow of blood across the specific site of the sample volume over time (systole). The cross-sectional area is calculated from the diameter of the LVOT (measured from the parasternal long-axis view) using the following formula:
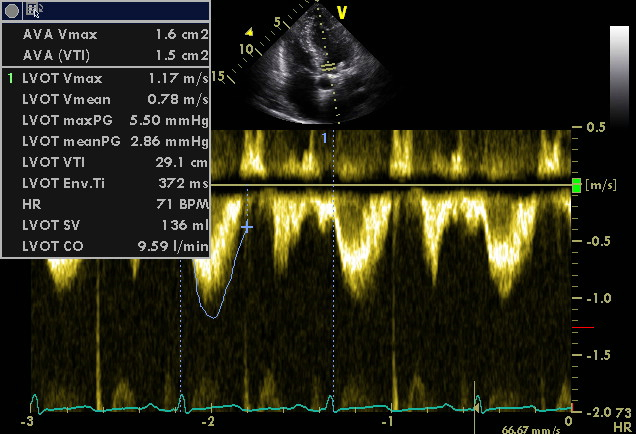
LVOT Area = π . (D/2)2
All you have to do now is multiply the area of the left ventricular outflow tract (LVOT area) by the velocity time integral (VTI). This will yield the so-called stroke volume (the quantity of blood expelled from the ventricle per beat). When you multiply stroke volume by heart rate you obtain cardiac output (the volume of ejected blood per minute). Finally, when you divide cardiac output by body surface area you obtain the cardiac index. Normal values at rest and exercise are shown in the table below.
| Rest | Exercise | |
|---|---|---|
| Stroke volume | 70 – 110ml | 80 – 130ml |
| Cardiac output | 5 – 8.5 L/min | 10 – 17 L/min |
| Cardiac index | > 2.5 L/min/m2 | > 5 L/min/m2 |
Stroke volume is also used to assess shunt flow and calculate aortic valve area using the continuity equation.
These hemodynamic parameters provide valuable information and help to detect the effects of treatment. There is only one major drawback: the calculations are largely dependent on the measurement of the left ventricular outflow tract. A small measurement error will result in large deviations in stroke volume (the radius of the outflow tract is squared). Nevertheless, hemodynamic measurements may provide incremental information, especially when you wish to observe the effects of treatment in individual patients. In relative terms, the size of the outflow tract is not important here because it does not change from exam to exam in one and the same patient. This allows you to detect subtle changes.
When you position your sample volume, make sure that your measurement of the LVOT diameter is as close as possible to the site at which you place the PW sample volume. Stroke volume measurements cannot be performed in the presence of high LVOT velocities (i.e. LVOT obstruction).
Stroke volume cannot be measured in several other situations as well.
Make sure you are truly in the LVOT and not in the aortic valve with your sample volume. The velocity in the aortic valve is usually higher.
The Doppler method is not the only technique to calculate stroke volume and its derivatives. Most scanners also calculate stroke volume on the basis of the Simpson method of ejection fraction or the MMode. This is done by subtracting the end-systolic volume from the end-diastolic volume. The difference should be the stroke volume. However, there is a major limitation: the method does not work in the presence of mitral regurgitation. This, and the fact that we tend to underestimate true volumes with echo, are the major limitations of this approach. Therefore it is rarely used.
Stroke volume can also be calculated from 2D echo and MMode measurements of volume. However, these methods are very inexact and should not be used.
3.2.3.2.5 Contractility - dP/dt
dP/dt is a parameter of myocardial contractility. It describes the rate of pressure rise in the left ventricle during systole. The faster the ventricle is able to „build up pressure, the better it functions. Thus, a rise in pressure can be derived from the CW Doppler spectrum of mitral regurgitation because velocities represent pressure gradients (by applying the Bernoulli equation of 4v2). As pressure in the left atrium during early systole may be neglected, the MR velocity is the instantaneous systolic pressure in the left ventricle (which resembles systolic blood pressure).
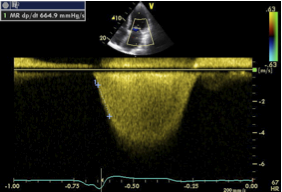
DP/dt is calculated from the slope of velocity between 1 and 3 m/sec by applying the following formula:
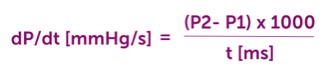
As we are measuring the slope between 1 and 3 m/sec (which represents the difference P2-P1 (36 – 4 mmHg), the formula may be simplified as follows: 32000/t
Normal values and the mode of grading severity are shown in the table below.
Limitations of the dP/dt method
Although the method is very appealing, it has several limitations:
- You need a good MR signal. This is obviously not always the case. Some patients have no MR signal while others have eccentric jets not optimally aligned to the CW.
- Even small differences in the time interval will lead to large differences in dP/dt (as one is dividing the time interval by a large number, i.e. 32000).
- The dP/dt method does not work in the presence of acute MR (LV pressure calculations are incorrect because of high left atrial pressures in acute MR).
- dP/dt is not entirely independent of loads. Therefore, alterations in preload or afterload affects the measurements.

| Normal | > 1200 mmHg/sec |
| Borderline | 800— 1200 mmHg/sec |
| Reduced | < 800 mmHg/sec |
| Severly reduced | < 500 mmHg/sec |
Limitations: MR signal needed, inexact, not completely load independent
You need a (good) MR CW Doppler spectral signal to measure dP/dt
3.2.3.2.6 Myocardial velocities and myocardial deformation parameters
With the help of tissue Doppler imaging (TDI) and speckle tracking echocardiography (STE) it is possible to directly obtain information about myocardial contractility. Basically these methods are used to assess myocardial velocities, strain, and strain rate. The principles of these, and the fundamentals of strain and strain rate imaging techniques are discussed in Chapter 1. These methods allow us to study the various components of contraction (radial, longitudinal and circumferential), and provide detailed information on global and regional systolic function.
3.2.3.2.6.1 Tissue Doppler Imaging (this aspect is partly addressed (blue) in Chapter 1)
Tissue Doppler is used to analyze the higher-amplitude, lower-velocity components of the Doppler signal which describe myocardial motion (see also Chapter 1). With TDI we can measure myocardial velocities within a region of interest (sample volume). The measurement is usually performed at the lateral mitral annulus or in the basal segments of the myocardium.
The major disadvantage of TDI is its “angle dependence”. In other words, we can only measure velocities parallel to the ultrasound beam. Thus, TDI is primarily used to measure longitudinal velocities from apical views. Typically the spectrum consists of an initially very sharp systolic signal (isovolumetric contraction), followed by the actual systolic peak (sm). In diastole we find two peaks which relate to early (Em) and late (Am) diastolic filling. To assess systolic function one should measure peak systolic velocity (sm), which has been shown to correlate with systolic function. Normal values are shown in the following table:
| Base | Apex | |
|---|---|---|
| Anterior | 6.5 ± 1.6 | 2.8 ±1 |
| Septal | 6.7 ± 1.4 | 2.8 ± 1.1 |
| Posterior | 6.5 ± 1.4 | 2.9 ± 1.3 |
| Lateral | 6.7 ± 1.5 | 3.2 ± 1.6 |
TDI can also be used to derive myocardial deformation parameters such as strain or strain rate. However these derivatives are angle dependent, moderately robust, and therefore rarely used in clinical practice. (Chapter 1)
TDI is a very simple way of looking at global systolic function. It has never gained widespread acceptance because it only measures left ventricular function in a specific area (where you place the sample volume). In addition, TDI cannot differentiate between active (contraction) and passive motion (translation and tethering effects). Furthermore, normal values for TDI velocities vary from segment to segment. Velocities general decrease as one moves towards the apex
Speckle tracking to assess systolic function
Strain and strain rate assessed by speckle tracking is a new approach. To review the methodology of deformation imaging / speckle tracking see section 1.9.
It is gaining acceptance rapidly and is now the preferred method to analyze deformation. In contrast to ejection fraction and hemodynamic measurements, it directly reveals the contractile function of the heart. As contractile dysfunction occurs before ejection fraction drops, it can be used as an early marker of systolic dysfunction. This is especially true for the longitudinal component of contraction, which can be studied from apical views. This has been shown to occur in numerous diseases including dilated cardiomyopathy, restrictive cardiomyopathy, left ventricular hypertrophy and valvular heart disease. Some pathologies show typical patterns of contractile dysfunction, both with respect to regional differences of strain and the components (longitudinal, circumferential and radial) of strain.
Longitudinal strain analysis showing a bull's eye image of the left ventricle. Global and regional longitudinal strain is normal:
Tracking of the myocardium for radial strain; red denotes contraction while blue denotes relaxation:
The parameter that best describes global systolic contractility is global longitudinal peak systolic strain (GLPSS). It is the average value of strain for the entire ventricle computed from all segments (apical views). Normal values are 18±2%.
Perform speckle tracking analysis in as many patients as possible. Although it not yet clear how this information translates into clinical practice, the method provides important insights. It is THE technology of the future.
Systolic dysfunction is frequently associated with abnormalities in the way the heart rotates during contraction. With the help of speckle tracking it is also possible to look at parameters of this motion, such as rotation and twist (see Chapter 1).
Limitations of speckle tracking:
Although speckle tracking is a promising technique, several issues remain unresolved. For instance, strain values differ between vendors (especially for circumferential and radial strain), normal values have not been firmly established, the tracking algorithms do not account for variations in myocardial thickness, and 2D speckle tracking does not consider "out of plane motion".
3.2.3.2.6.3 Assessment of Strain - Practical Issues
Speckle tracking analysis is an offline method. It requires you to first record the digital image loops. Dedicated software then performs a semiautomated process where the myocardium is traced and tracked throughout the cardiac cycle.
The software then calculates strain and other deformation parameters and also sets the time of aortic valve closure. You can also manually adjust the time of aortic valve closure by using Doppler methods (LVOT signal). The results are displays in various formats. For example in the form of a „bulls eye display“ where the strain values of the entire heart are color encoded, as curves that show the change in strain (or strain rate) over time or as in the format of an anatomical MMode.
When you acquire the loops make sure that the entire ventricle is within the sector. This can be difficult at times, especially if the ventricle is very large. The region of interest should be optimized so that as much of the myocardium (but not the pericardium) is included. There will always be a compromise but improper myocardial delineation is a major source of error. Make sure that the cycle length of all views are approximately the same and avoid ectopic and post ectopic beats. The method can also be applied in atrial fibrillation, however expect larger variations between different beats. In addition, you will have to look at each view individually since variations in the cycle length will not permit a bulls eye display. The method is fairly robust and also works well in situations where image quality is not the best. Still, strive for the best image quality possible.
Longitudinal strain might be used as a screening method to detect abnormalities of contraction.